|
coordinating the naming of a new element, involving the research laboratory and allowing for public comments.assessing claims resulting in the validation and assignation of an element discovery.defining the structure of a temporary name and symbol.establishing the criteria for a new element discovery.In particular, IUPAC is directly involved in the following: IUPAC involvement covers various aspects of the table and data that it unveils, and several reports and recommendations, some quite recent, attest of that input. – Access at a full resolution of this Table as PDF (made available by King’s Center for Visualization in Science).īy virtue of its work in relation with the chemical elements, IUPAC can dispense a periodic table that is up-to-date. – Review the latest IPTEI element-by-element review including a chart of all known stable and radioactive isotopes for each element and examples practical applications of isotopic measurements and technologies – Explore the interactive version at /isotopes-matter (or see release) – Read “Atomic Weights: No Longer Constants of Nature”, Chem Int 33(2), 10–15 (2011), IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Educational Community Coplen, Peter Mahaffy, Ian Mills, Roberto Marquardt, and more. Leigh, Sigurd Hofmann, Eric Scerri, Juris Meija, Norman E. See PAC (AOP ) for full details or visit Commission II.1 ĭownload the PDF version (letter size or A4) or A3 (PDF) or see earlier versionsĬheck out SPECIAL Chem Int Jan 2019 - International Year of the Periodic Table (IYPT) - with contributions by Jan Reedijk, Natalia Tarasova, G.J. 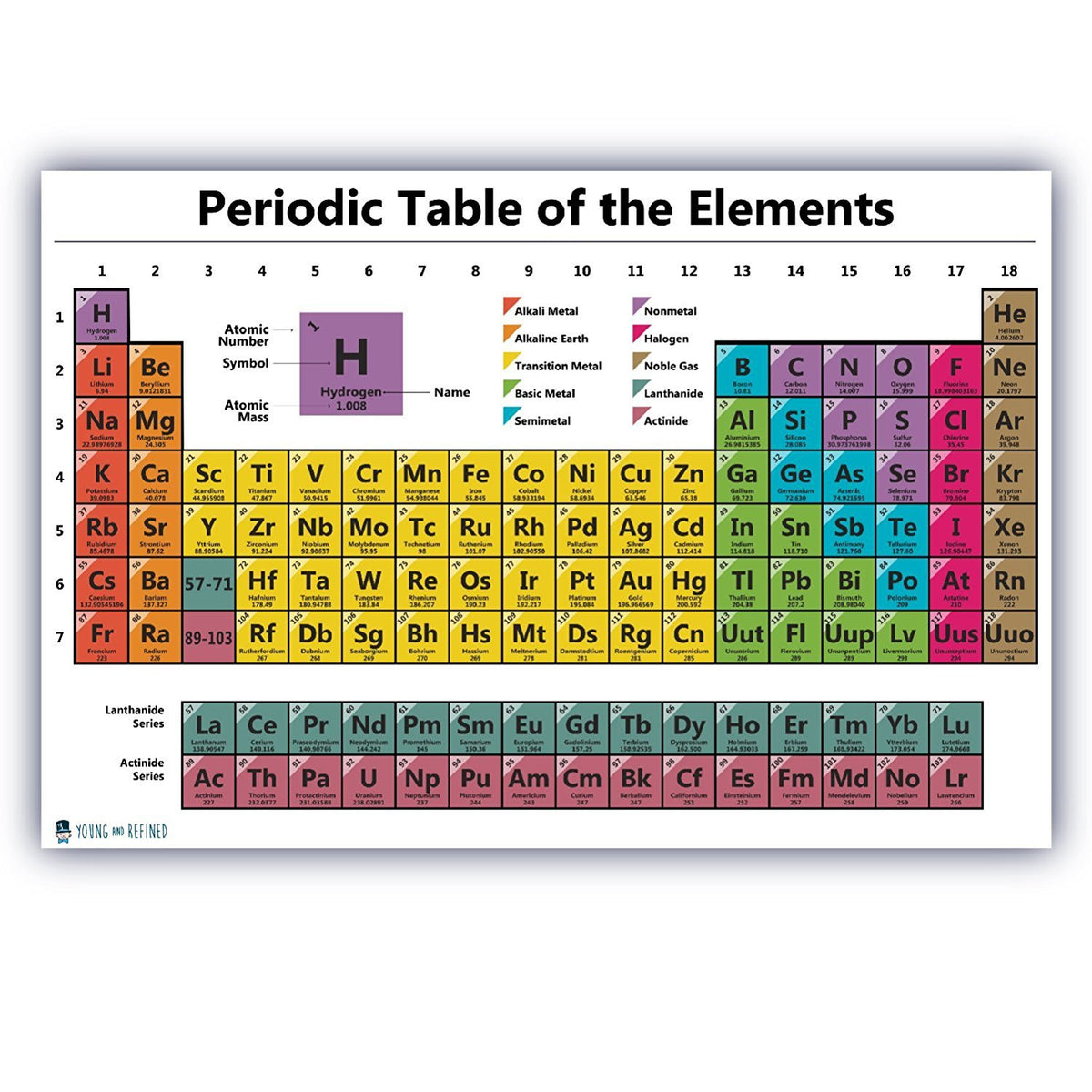
For elements that lack isotopes with a characteristic isotopic abundance in natural terrestrial samples, the mass number of the nuclide with the longest confirmed half-life is listed between square brackets. Unfortunately, there was a slightly different system in place in Europe.The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW), compiled as part of the 2021 Table of Standard Atomic Weights 2021. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. 
Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. 
A new period begins when a new principal energy level begins filling with electrons. There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
